Mesenchymal Stromal Cells: A Comprehensive Guide to Research and Therapy
Mesenchymal Stromal Cells (MSCs) keep showing why they matter in regenerative medicine and advanced therapies. We see a blend of multipotent differentiation and strong immunomodulatory effects that maps to real clinical needs. From isolation to therapeutic use, getting the details right moves research forward. Here we cover the key aspects of MSC biology and the ways they are being put to work.
Understanding Mesenchymal Stromal Cells
Mesenchymal Stromal Cells (MSCs) are multipotent stromal cells that can differentiate into osteocytes, chondrocytes, and adipocytes. We recognize their central role in tissue repair, regeneration, and immunomodulation. These cells are typically isolated from bone marrow, adipose tissue, and umbilical cord blood. Their biological characteristics make them well suited for both research and therapy. Our understanding continues to grow, opening new directions for their use in medicine.
1. Defining MSCs and Their Unique Properties
MSCs are non-hematopoietic, multipotent stromal cells characterized by their adherence to plastic in vitro. We identify them by specific surface markers, including CD73, CD90, and CD105, while lacking hematopoietic markers like CD34, CD45, and HLA-DR. These cells possess notable self-renewal capacity and can differentiate into mesenchymal lineages. Their immunomodulatory properties, such as secreting various trophic factors and cytokines, enable them to regulate immune responses and promote tissue healing. This combination of properties underpins their value in regenerative medicine.
2. Historical Perspective and Evolving Terminology
The concept of MSCs originated from the discovery of fibroblast-like cells in bone marrow by Friedenstein in the 1970s. Initially termed “colony-forming unit-fibroblasts” (CFU-F), their multipotent nature was later recognized. The term “Mesenchymal Stem Cell” gained popularity, but due to ongoing debates about their true stem cell characteristics in vivo, the International Society for Cellular Therapy (ISCT) proposed “Mesenchymal Stromal Cell.” This revised terminology emphasizes their stromal origin and functional properties rather than strict stemness. We continue to refine our understanding of these cells.
Isolation, Characterization, and Expansion of MSCs
Effective isolation, rigorous characterization, and efficient expansion are foundational for successful MSC research and therapeutic development. We employ standardized protocols to ensure consistent, high-quality cell populations. The choice of isolation source influences MSC characteristics and downstream use. Our methods prioritize cell viability and potency throughout each step.
Isolation Sources and Methods
MSCs can be isolated from various tissues. Bone marrow is a traditional source, yielding MSCs with robust differentiation potential. Adipose tissue provides a more abundant source, often with easier access. Umbilical cord tissue and Wharton’s jelly are also rich sources, offering a less invasive collection method and lower immunogenicity. We typically isolate MSCs using density gradient centrifugation followed by plastic adherence.
Characterization and Quality Control
After isolation, we characterize MSCs based on three minimal criteria established by the ISCT: plastic adherence, specific surface marker expression (CD73+, CD90+, CD105+, and CD34-, CD45-, HLA-DR-), and multipotent differentiation potential (osteogenic, chondrogenic, and adipogenic). Flow cytometry confirms surface marker expression. In vitro differentiation assays verify multipotency. These stringent characterization steps ensure the identity and purity of our MSC populations.
Expansion and Culture Conditions
We expand MSCs in vitro to obtain sufficient cell numbers for research or clinical applications. Culture conditions, including media composition, growth factors, and oxygen levels, are critical for maintaining MSC properties. Serum-free and xeno-free media are increasingly preferred to enhance safety and reduce variability for clinical use. We utilize specific recombinant proteins to optimize cell proliferation and prevent premature differentiation.
Here is a comparison of common MSC isolation sources:
| Source | Advantages | Disadvantages | Typical Yield (cells/g tissue) |
|---|---|---|---|
| Bone Marrow | Well-established, high differentiation | Invasive collection, donor variability | $10^3 – 10^4$ |
| Adipose Tissue | Abundant, less invasive collection | Heterogeneous population, lower proliferation | $10^4 – 10^5$ |
| Umbilical Cord | Non-invasive, immunoprivileged, high yield | Less established clinical use | $10^5 – 10^6$ |
Therapeutic Potential and Clinical Applications of MSCs
The therapeutic potential of MSCs stems from their multifaceted biological functions. We observe their ability to modulate immune responses, secrete trophic factors, and contribute to tissue repair. These properties position MSCs as a promising therapeutic agent for a wide spectrum of diseases. Clinical trials are actively assessing their efficacy across multiple conditions.
Immunomodulation and Anti-inflammatory Effects
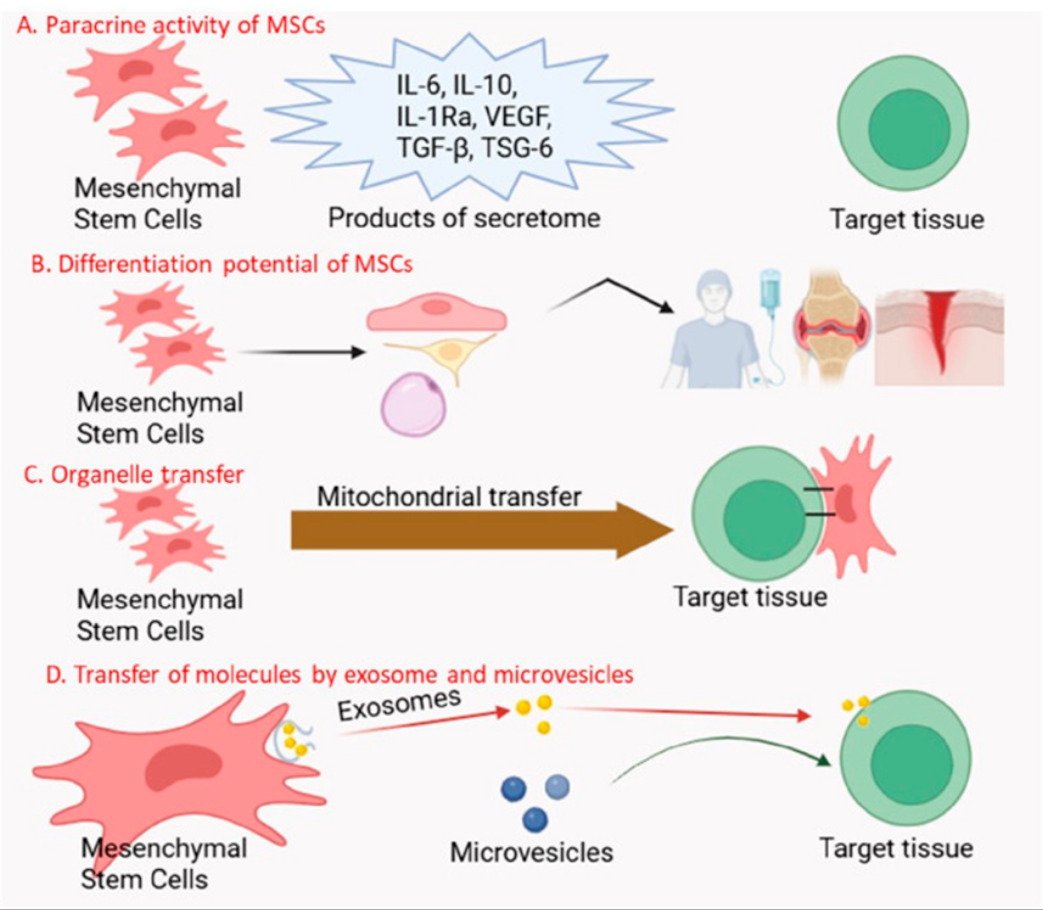
MSCs exert potent immunomodulatory effects by interacting with various immune cells, including T cells, B cells, NK cells, and dendritic cells. They secrete numerous soluble factors, such as PGE2, TGF-β, and IL-10, which suppress immune cell proliferation and cytokine production. This makes MSCs valuable for treating autoimmune diseases, graft-versus-host disease (GVHD), and inflammatory conditions. We leverage these properties in developing novel therapeutic strategies.
Tissue Repair and Regeneration
MSCs contribute to tissue repair through several mechanisms. They can differentiate into specialized cells, directly replacing damaged tissue. Additionally, they release trophic factors that promote angiogenesis, reduce apoptosis, and stimulate endogenous progenitor cells. These effects are vital for regenerating damaged tissues in conditions like myocardial infarction, osteoarthritis, and neurological disorders. We focus on enhancing these regenerative capacities.
Clinical Trials and Future Prospects
Numerous clinical trials are currently investigating MSC-based therapies for diverse indications. These include cardiovascular diseases, orthopedic injuries, neurological disorders, and autoimmune conditions. While promising results have emerged, challenges remain in standardizing manufacturing processes and defining optimal dosing regimens. We anticipate that ongoing research will further refine their clinical application.
MSCs in Biomanufacturing and Advanced Cell Therapies
The successful translation of MSCs from research to clinical use depends on robust biomanufacturing. We focus on scalable, cost-effective methods that yield high-quality MSCs. This includes optimizing culture conditions and ensuring product consistency. Our experience with recombinant protein production directly supports these advanced cell therapies.
Large-Scale Production and Process Optimization
Scaling up MSC production involves transitioning from traditional 2D culture systems to 3D bioreactors. We optimize parameters such as cell seeding density, media perfusion rates, and oxygen tension to maximize cell yield and maintain potency. Automation and closed systems are integrated to reduce contamination risks and improve process efficiency. These advances are key to meeting clinical demand.
Quality Control and Regulatory Compliance
Maintaining stringent quality control is paramount for therapeutic MSC products. We run a full panel of assays to assess cell viability, purity, identity, and potency. Endotoxin testing and mycoplasma detection are routinely conducted to ensure product safety. Compliance with Good Manufacturing Practice (GMP) guidelines is required for regulatory approval. We ensure our processes meet these rigorous standards.
Role of Recombinant Proteins in MSC Culture
Recombinant proteins are indispensable for developing defined, serum-free, and xeno-free media for MSC culture. These proteins, including FGFs like Recombinant Human FGF-2/bFGF and GFs such as Recombinant Human IL-6, provide necessary growth factors and cytokines. They promote MSC proliferation, maintain multipotency, and support specific differentiation pathways. Using high-purity recombinant proteins minimizes variability and enhances the safety profile of cell therapy products. We provide a range of high-quality recombinant proteins for these applications.
Future Directions and Innovations in MSC Research
The field of MSC research is rapidly evolving, with new ideas moving quickly from concept to testing. We are exploring strategies to raise therapeutic efficacy and broaden applications. These advancements promise to bring more of the potential of MSC-based therapies into practice. Our research initiatives contribute to this dynamic landscape.
Exosomes and Cell-Free Therapies
MSCs exert many of their therapeutic effects through the secretion of extracellular vesicles, particularly exosomes. These nanovesicles contain proteins, lipids, and nucleic acids that can modulate recipient cell function. Research is focusing on developing exosome-based therapies, offering a cell-free alternative to direct MSC transplantation. This approach could address challenges linked to cell delivery and persistence.
Gene Editing and Enhanced MSC Function
Advances in gene editing technologies, such as CRISPR-Cas9, allow for precise modification of MSCs. We can engineer MSCs to overexpress specific trophic factors, enhance immunomodulatory properties, or resist immune rejection. This genetic engineering holds the potential to create “super-MSCs” with improved therapeutic efficacy for targeted applications. We investigate these advanced approaches.
Organoids and Disease Modeling
MSCs are increasingly utilized in the development of 3D organoid models. These complex in vitro systems better mimic in vivo tissue architecture and function compared to traditional 2D cultures. Organoids incorporating MSCs can serve as powerful tools for disease modeling, drug screening, and understanding tissue development. We support research in this innovative area.
Partner with East-Mab Bio for Your Mesenchymal Stromal Cell Research and Development
Advance your MSC research and therapeutic development with high-quality recombinant protein raw materials from Jiangsu East-Mab Biomedical Technology Co., Ltd. Our world-class platform and commitment to excellence ensure you have the reliable components needed for consistent, scalable, and impactful results. Explore our range of cell culture proteins and diagnostic enzymes, or contact our experts to discuss your specific needs.
Contact us today to learn more about how East-Mab Bio can support your advancements in cell therapy and regenerative medicine.
Email: product@eastmab.com
Phone: +86-400-998-0106
| Name | Cat# | Species | Express Host | Purity | Endotoxin (EU/mg) |
| Laminin 521 E8 | Y13342H | Human | HEK293 | ≥95% | ≤10 |
| Laminin 511 E8 | Y13242H | Human | HEK293 | ≥95% | ≤10 |
| Vitronectin | Y04842/L | Human | HEK293 | ≥95% | ≤10 |
| Fibronectin | Y13142H | Human | HEK293 | ≥95% | ≤10 |
| bFGF | Y00351H | Human | E. coli | ≥95% | ≤10 |
| TGF-β1 | Y04201N | Human | CHO | ≥95% | ≤10 |
| TGF-β3 | Y07901 | Human | CHO | ≥95% | ≤10 |
| LR3-IGF1 | Y03101 | Human | CHO | ≥95% | ≤10 |
| VEGF165 | Y04701 | Human | CHO | ≥95% | ≤10 |
| BMP-2 | Y00421H | Human | E. coli | ≥95% | ≤10 |
| EGF | Y00801 | Human | CHO | ≥95% | ≤10 |
| PDGF-BB | Y03411 | Human | CHO | ≥95% | ≤10 |
What are the primary sources for isolating Mesenchymal Stromal Cells?
MSCs can be isolated from various tissues, with bone marrow, adipose tissue, and umbilical cord being the most common and well-studied sources. Each source offers unique advantages and considerations regarding cell yield, differentiation potential, and immunomodulatory properties. Other sources include dental pulp, synovial membrane, and peripheral blood, though these are less frequently used for clinical applications.
How do Mesenchymal Stromal Cells contribute to regenerative medicine?
MSCs contribute to regenerative medicine through multiple mechanisms, including their ability to differentiate into various cell types (e.g., osteocytes, chondrocytes, adipocytes), secrete trophic factors that promote tissue repair and angiogenesis, and exert potent immunomodulatory and anti-inflammatory effects. These properties make them valuable for treating conditions involving tissue damage, inflammation, and immune dysregulation.
What are the main challenges in the large-scale manufacturing of MSCs for clinical use?
Large-scale manufacturing of MSCs presents several challenges, including ensuring cell consistency and quality across batches, optimizing culture conditions for expansion while maintaining therapeutic potency, developing serum-free and xeno-free media, and addressing regulatory hurdles for good manufacturing practice (GMP) compliance. Sourcing high-quality and consistent raw materials, such as recombinant proteins, is critical to overcoming these challenges.
What role do recombinant proteins play in Mesenchymal Stromal Cell culture?
Recombinant proteins are essential components in modern MSC culture media, particularly in serum-free and xeno-free formulations. They provide defined growth factors, cytokines, and attachment factors that are critical for MSC proliferation, viability, and maintenance of multipotency. Using high-quality recombinant proteins ensures consistency, reduces variability, and enhances the safety profile of MSC products for clinical applications.