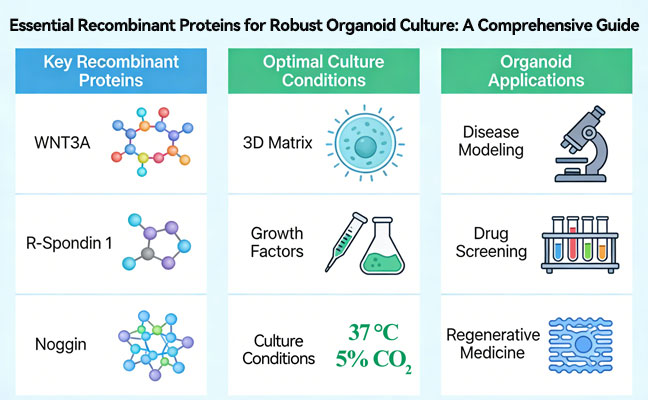
Recombinant Mouse G-CSF: Function, Applications, and Quality
Recombinant mouse G-CSF sits at the center of a lot of myeloid biology work, and for good reason. This cytokine does exactly what researchers need when studying neutrophil development, stem cell mobilization, or immune function in mouse models. The challenge has always been getting material that performs consistently across different experimental setups. What follows covers the biological mechanisms, practical applications, and quality factors that determine whether your recombinant mouse G-CSF actually delivers reliable results.
How Recombinant Mouse G-CSF Works at the Molecular Level
Granulocyte colony-stimulating factor functions as a glycoprotein that controls granulocyte production, differentiation, and activity. Neutrophils respond most directly to this signal. In mice, G-CSF maintains baseline innate immunity and ramps up neutrophil output during infection. The cytokine works through G-CSFR, its dedicated receptor found on myeloid progenitors and mature neutrophils. Receptor binding triggers a cascade of intracellular events that ultimately change gene expression patterns.
JAK1 and JAK2 activation represents the first major signaling step. These Janus kinases phosphorylate the G-CSFR cytoplasmic domain, which then recruits STAT proteins. STAT3 predominates in this pathway. Once phosphorylated, STAT3 molecules pair up, move into the nucleus, and bind DNA sequences that control proliferation, differentiation, and survival genes. This JAK-STAT signaling forms the backbone of neutrophil production.
G-CSF also engages the MAPK pathway through ERK1/2, contributing to proliferative and anti-apoptotic effects. The PI3K/Akt pathway handles cell survival signaling, preventing programmed death in developing myeloid cells. These pathways work together to maintain robust granulopoiesis. Recombinant mouse G-CSF replicates these natural functions, giving researchers precise control over myeloid cell development in both normal physiology studies and disease models.
What Mouse G-CSF Actually Does In Vivo and In Vitro
The primary in vivo effect is straightforward: G-CSF pushes the bone marrow to produce and release neutrophils. Circulating neutrophil counts rise substantially. This matters for infection models and myelosuppression studies where neutrophil recovery needs tracking. G-CSF also mobilizes hematopoietic stem cells from marrow into peripheral blood, which becomes important for transplantation research.
In vitro applications center on cell culture protocols supporting myeloid progenitor growth and differentiation. Recombinant mouse G-CSF becomes essential for granulopoiesis assays and neutrophil maturation studies. Researchers use it to drive stem cell differentiation toward granulocytic lineages, generating insights into developmental biology and immune cell function. These controlled conditions allow detailed investigation of cellular responses that would be difficult to isolate in whole-animal systems.
Where Recombinant Mouse G-CSF Gets Used in Research
Oncology research uses G-CSF to manage chemotherapy-induced neutropenia in mouse models. This allows testing of higher anti-cancer drug doses without losing animals to infection. Researchers can evaluate novel chemotherapies while controlling for immunosuppression effects that would otherwise confound results. The cytokine essentially separates drug toxicity from immune compromise.
Regenerative medicine applications focus on stem cell mobilization and collection for transplantation studies. Recombinant mouse G-CSF increases the yield of hematopoietic stem cells available for harvest and subsequent use in tissue repair experiments. In vitro expansion protocols also depend on G-CSF to maintain stem cell populations during culture.
Immunology labs rely on mouse G-CSF to generate neutrophils for functional assays. Phagocytosis studies, chemotaxis experiments, and inflammation models all require consistent neutrophil supplies. The cytokine also supports organoid development where myeloid components need to differentiate properly within complex cellular structures. Diagnostic assay development uses recombinant G-CSF as a raw material for cell-based platforms.
| Application Area | Research Focus | Key Benefit of Mouse G-CSF |
|---|---|---|
| Oncology | Chemotherapy-induced neutropenia, drug discovery | Mitigates myelosuppression, enables higher drug doses |
| Regenerative Medicine | Stem cell mobilization, tissue repair | Enhances stem cell availability for transplantation and culture |
| Immunology | Neutrophil function, inflammation, immunomodulation | Facilitates neutrophil generation for assays, models immune responses |
| Organoid Development | Disease modeling, developmental biology | Supports differentiation and growth of myeloid components |
| In Vitro Diagnostics | Assay development, quality control | Critical raw material for cell-based diagnostic platforms |
| Cell Therapy Research | Cell expansion, differentiation | Promotes specific cell lineage development for therapeutic use |
Getting Mouse G-CSF to Work in Cell Culture and IVD Systems
Serum-free media formulations often include recombinant mouse G-CSF to support hematopoietic cell proliferation and differentiation. Defined conditions improve reproducibility compared to serum-containing systems. Optimal concentrations vary by cell line and application, so empirical determination remains necessary. Sterility and proper handling prevent contamination and protein degradation.
IVD platforms use recombinant mouse G-CSF as controls or calibrators in diagnostic kits. High purity and consistent biological activity directly affect diagnostic accuracy. Batch-to-batch consistency becomes critical when diagnostic results need to meet regulatory standards. Comprehensive documentation supports compliance requirements.
What Makes Recombinant Mouse G-CSF Reliable
Purity and biological activity determine whether recombinant mouse G-CSF performs as expected. Production quality control starts with expression system selection. Mammalian cells like CHO provide proper post-translational modifications that affect protein folding and function. Bacterial expression systems can produce functional protein but may lack glycosylation patterns that influence stability and activity.
Purification typically involves multiple chromatography steps to achieve purity above 95% as measured by SDS-PAGE and HPLC. Endotoxin control matters significantly for cell culture and in vivo applications. Levels below 1 EU/mg prevent non-specific immune activation that would confound experimental results. Bioactivity testing using G-CSF-dependent cell lines confirms that purified protein actually stimulates the expected cellular responses.
East-Mab maintains these standards through a production platform backed by over $30 million in investment. Our quality control includes purity assessment, endotoxin testing, and stability studies that verify batch consistency over time.
| Quality Parameter | East-Mab Standard | Importance for Research |
|---|---|---|
| Purity | ≥95% (SDS-PAGE, HPLC) | Minimizes interference from host cell proteins |
| Endotoxin Levels | ≤1 EU/mg | Prevents non-specific immune responses, ensures cell viability |
| Biological Activity | Verified by specific cell proliferation bioassays | Confirms functional efficacy and expected physiological response |
| Expression System | Mammalian (e.g., CHO) | Ensures proper folding and post-translational modifications |
| Stability | Long-term stability data available | Guarantees consistent performance over time |
| Batch Consistency | Rigorous batch-to-batch QC | Ensures reproducibility of experimental results |
How East-Mab Produces Recombinant Mouse G-CSF
Jiangsu East-Mab Biomedical Technology Co., Ltd. focuses on recombinant protein raw materials using state-of-the-art expression and purification technologies. Each batch undergoes protein validation including detailed purity analysis, endotoxin testing, and bioactivity assays. This approach ensures products meet the demanding requirements of biomedical research applications.
Finding a Supplier That Delivers Consistent Results
Supplier selection affects experimental reproducibility more than many researchers realize. Reputation and manufacturing experience indicate whether a supplier can actually produce consistent material. East-Mab transitioned from antibody drug development to recombinant protein raw materials, bringing process expertise to cytokine production.
Technical support availability matters when troubleshooting experimental issues or interpreting product specifications. Batch consistency documentation, including certificates of analysis and safety data sheets, provides transparency about what you’re actually receiving. Production standards that approach GMP practices generally correlate with better product quality.
FAQs
What does mouse G-CSF actually do biologically?
Mouse G-CSF stimulates neutrophil production, maturation, and release from bone marrow. The cytokine supports innate immune function by increasing circulating neutrophil counts during infection. It also mobilizes hematopoietic stem cells into peripheral blood.
How do manufacturers verify biological activity?
Cell proliferation assays using G-CSF-dependent lines like NFS-60 cells provide the standard measurement. Cells are cultured with varying G-CSF concentrations, and proliferation is quantified to determine ED₅₀ values. This indicates the concentration needed for half-maximal stimulation.
Why does endotoxin level matter so much?
Endotoxins from bacterial cell walls trigger inflammatory responses and non-specific cellular activation. High endotoxin in cell culture confounds results by activating pathways unrelated to G-CSF signaling. In vivo applications with high endotoxin material can cause severe adverse reactions in animals.
Does recombinant mouse G-CSF work in serum-free systems?
Yes, and serum-free conditions often improve experimental reproducibility. G-CSF provides defined growth factor stimulation without the variability that serum introduces. This creates more controlled conditions for studying specific cellular responses.
What advantages come from using a specialized supplier?
Specialized suppliers like East-Mab produce high-purity, high-activity recombinant proteins under stringent quality control. Batch-to-batch consistency, low endotoxin levels, and verified biological activity support reproducible research. Technical support and comprehensive documentation add practical value.
Partner with East-Mab for Your Recombinant Protein Needs
For high-quality recombinant mouse G-CSF and other recombinant protein raw materials, partner with Jiangsu East-Mab Biomedical Technology Co., Ltd. Our world-class production platform and rigorous quality assurance protocols ensure you receive products of exceptional purity and biological activity. Contact us today to discuss your research or diagnostic needs. Reach us at +86-400-998-0106 or product@eastmab.com.