T Cell Culture Guide: Principles, Techniques, and Applications
Getting T cell culture right sits at the heart of progress in immunology and cell therapy. From experience, a solid grasp of T cell biology paired with careful hands-on work makes the difference. Time and again, we see that high-quality recombinant proteins are non-negotiable for reliable, reproducible outcomes in these complex systems. Below, we walk through the essential principles and techniques.
Understanding T Cell Culture Fundamentals
T cell culture involves growing and manipulating T lymphocytes outside their natural environment. This process is vital for understanding immune responses, developing new therapies, and conducting drug screening. T cells are central to adaptive immunity, recognizing and eliminating pathogens or abnormal cells. Their diverse roles include direct killing (cytotoxic T cells), coordinating immune responses (helper T cells), and regulating immune activity (regulatory T cells).
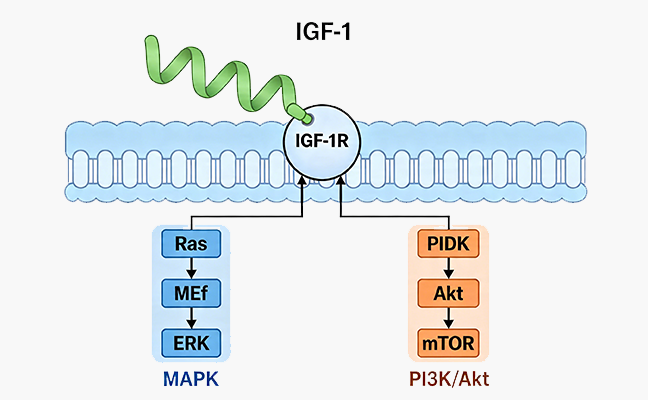
Successful T cell culture relies on carefully controlled conditions that mimic the physiological environment necessary for T cell survival, proliferation, and function. Key components include a basal medium providing essential nutrients, amino acids, and vitamins. Serum or serum substitutes supply growth factors and other necessary proteins. Additionally, specific recombinant proteins, such as cytokines, drive T cell growth and differentiation. For instance, recombinant human IL-2 supports general T cell proliferation, while IL-7 and IL-15 are important for T cell survival and homeostatic expansion. The quality and consistency of these raw materials directly impact experimental outcomes.
The Biology of T Cells and Their Role
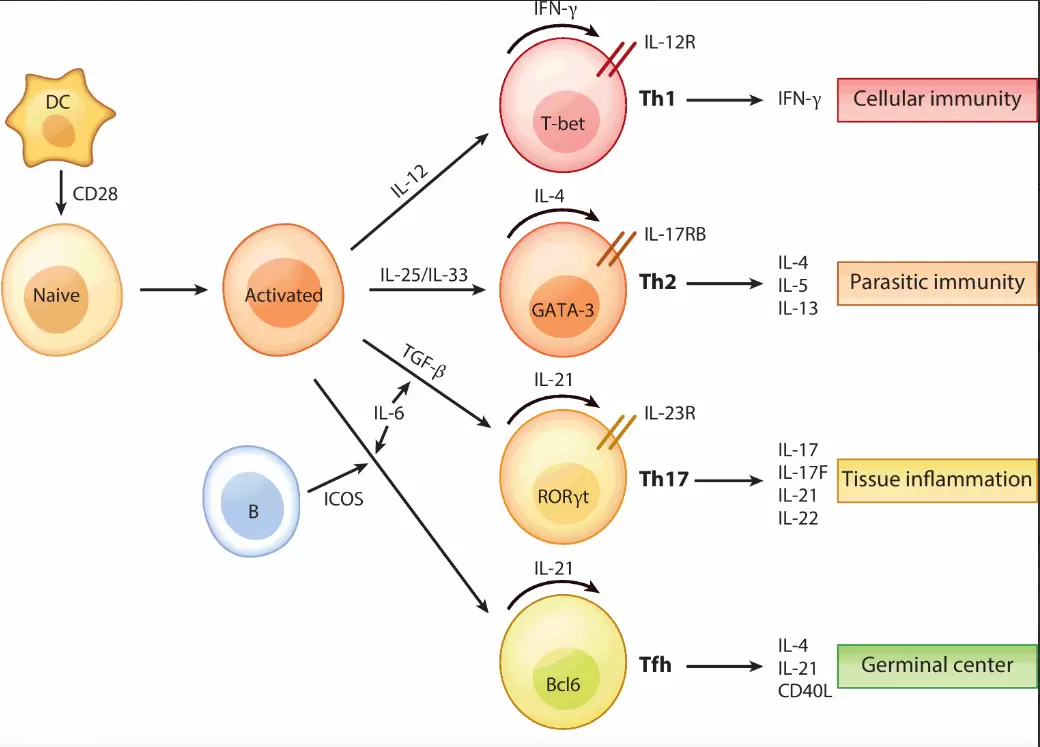
T cells originate in the bone marrow and mature in the thymus. They express a T cell receptor (TCR) that recognizes specific antigens presented by major histocompatibility complex (MHC) molecules. This antigen recognition, coupled with co-stimulatory signals, activates T cells. Activated T cells then proliferate and differentiate into effector cells. These effector cells perform various functions, including direct cytotoxicity, cytokine secretion, and memory formation. Understanding these biological processes is essential for designing effective culture strategies.
Key Components of T Cell Culture Media
T cell culture media typically include a basal medium, serum or serum-free supplements, and various recombinant proteins. Basal media provide the fundamental nutritional requirements. Serum, often fetal bovine serum (FBS), supplies a complex mixture of growth factors and hormones. However, serum-free media are increasingly preferred for clinical applications due to reduced variability and contamination risks. Recombinant cytokines, such as recombinant human IL-2, IL-4, IL-6, IL-7, IL-10, IL-12, IL-13, and IL-15, are frequently added to promote specific T cell activities. These recombinant proteins ensure defined and reproducible culture conditions.
Essential Techniques for Successful T Cell Culture
Achieving robust T cell expansion requires meticulous technique, from initial cell isolation to optimized growth conditions. We prioritize methods that ensure high viability and functionality. Our protocols emphasize aseptic practices and the use of high-quality reagents. This approach minimizes variability and supports consistent experimental results.
Isolation and Activation of T Cells
T cells can be isolated from various sources, including peripheral blood mononuclear cells (PBMCs) and apheresis products. Density gradient centrifugation or magnetic bead separation are common isolation methods. Once isolated, T cells require activation to induce proliferation. This typically involves stimulating the TCR and a co-stimulatory receptor. Anti-CD3 and anti-CD28 antibodies, often immobilized on beads, are widely used for this purpose. This dual stimulation mimics physiological T cell activation.
Optimizing T Cell Expansion and Growth
Optimal T cell expansion depends on several factors, including cell density, media composition, and the presence of appropriate growth factors. We maintain cell densities within a specific range to prevent overgrowth or nutrient depletion. Regular media changes are crucial for replenishing nutrients and removing waste products. The addition of key recombinant cytokines is critical. For example, Recombinant Human IL-2 is required for T cell proliferation, while IL-7 and IL-15 promote long-term survival and memory T cell development. These factors ensure sustained and healthy cell growth.
Applications of T Cell Culture in Research and Therapy
T cell culture is a cornerstone of modern biomedical research and therapeutic development. We leverage its versatility across various applications. Our work contributes to both fundamental immunological understanding and the advancement of cutting-edge treatments.
T Cells in Immunotherapy Development
T cell culture is indispensable for developing T cell-based immunotherapies. These therapies harness the body's immune system to fight diseases like cancer. Researchers culture T cells to study their anti-tumor activity and engineer them for enhanced targeting. This includes developing novel T cell receptors or chimeric antigen receptors (CARs). The ability to expand and manipulate T cells ex vivo is central to these therapeutic strategies.
Advancements in CAR-T and TCR-T Cell Therapies
CAR-T and TCR-T cell therapies represent significant breakthroughs in cancer treatment. These approaches involve isolating a patient's T cells, genetically modifying them in culture to recognize cancer-specific antigens, and then reinfusing them. The modified T cells proliferate and target tumor cells. Robust T cell culture methods are essential for the large-scale expansion of these engineered cells. This ensures sufficient cell numbers for effective clinical application.
Overcoming Challenges in T Cell Culture
T cell culture presents several challenges, including maintaining cell viability, preventing contamination, and ensuring consistent results. We address these issues through optimized protocols and stringent quality control. Our experience highlights the importance of high-quality raw materials in mitigating these hurdles.
Maintaining T cell viability and functionality over extended periods is crucial for many applications. This requires careful control of culture conditions, including pH, temperature, and nutrient availability. Contamination, particularly by mycoplasma, can compromise experimental integrity. Adhering to strict aseptic techniques and routine testing are essential preventive measures. Variability between donors or reagent batches can also impact reproducibility. Using GMP-grade recombinant proteins and defined, serum-free media helps standardize culture conditions, reducing batch-to-batch inconsistencies. Our recombinant protein products, such as our range of FGFs, are manufactured to high purity standards to support reliable T cell expansion.
Future Directions and Innovations in T Cell Culture
The field of T cell culture is rapidly evolving, driven by technological advancements and increasing therapeutic demands. We are actively involved in exploring and implementing these innovations. Our focus remains on enhancing efficiency, scalability, and therapeutic efficacy.
New media formulations are being developed to support specific T cell subsets and functions. Advanced bioreactor systems enable large-scale, automated T cell expansion, crucial for clinical manufacturing. Automation and high-throughput screening technologies streamline experimental workflows and accelerate discovery. Gene editing tools, such as CRISPR, offer unprecedented opportunities to engineer T cells with enhanced anti-tumor activity or reduced off-target effects. These innovations pave the way for more personalized and precision T cell therapies. Continuous innovation in recombinant protein raw materials, offering improved stability and bioactivity, will be critical enablers for these future developments.
| Name | Cat# | Species | Expression System | Purity | Endotoxin |
IL-2 | Y02001N | Human | CHO | ≥95% | ≤1 EU/mg |
| IL-4 | Y02201 | Human | CHO | ≥95% | ≤10 EU/mg |
| IL-7 | Y02401 | Human | CHO | ≥95% | ≤10 EU/mg |
| IL-12 | Y04001 | Human | CHO | ≥95% | ≤10 EU/mg |
| IL-15 | Y02701 | Human | CHO | ≥95% | ≤10 EU/mg |
| IL-21 | Y02901 | Human | CHO | ≥95% | ≤10 EU/mg |
| Anti-Human CD3 mAb | C00404 | Human | CHO | ≥95% | ≤1 EU/mg |
| Anti-Human CD28 mAb | C02302 | Human | CHO | ≥95% | ≤1 EU/mg |
| IFN-γ | Y01801 | Human | CHO | ≥95% | ≤10 EU/mg |
| IFN-γ | Y01821H | Human | E. coli | ≥95% | ≤10 EU/mg |
Frequently Asked Questions
What are the most critical components for successful T cell culture?
Successful T cell culture relies on several critical components, including a suitable basal medium, serum or serum substitutes, growth factors (e.g., IL-2, IL-7, IL-15), T cell activators (e.g., anti-CD3/CD28 antibodies), and antibiotics. High-quality recombinant proteins are indispensable for consistent and reliable results.
How can I prevent contamination in my T cell cultures?
Preventing contamination requires strict aseptic technique, working in a sterile laminar flow hood, using sterile reagents and consumables, regularly cleaning incubators, and performing routine mycoplasma testing. Using antibiotics in media can help but should not replace good aseptic practices.
What are the main applications of T cell culture in immunotherapy?
T cell culture is fundamental to developing immunotherapies like CAR-T and TCR-T cell therapies, where T cells are genetically engineered and expanded ex vivo to target cancer cells. It’s also used for vaccine development, immune monitoring, and studying T cell responses to various diseases.
What is the difference between primary T cells and T cell lines in culture?
Primary T cells are isolated directly from biological samples (e.g., blood) and have limited lifespan and expansion capacity in vitro, maintaining closer physiological relevance. T cell lines are immortalized cells that can proliferate indefinitely but may have altered characteristics compared to primary cells. Both have specific applications in research.
Why is the quality of recombinant proteins important for T cell culture?
The quality of recombinant proteins (e.g., cytokines, growth factors) matters because they directly influence T cell viability, proliferation, and functionality. High-purity, low-endotoxin, and consistent recombinant proteins ensure reproducible experimental results and are essential for clinical-grade cell therapy manufacturing.
Elevate Your T Cell Culture with East-Mab Biomedical
Discover how Jiangsu East-Mab Biomedical Technology Co., Ltd.'s high-quality recombinant protein raw materials can strengthen your T cell culture research and therapeutic development. Contact us today to learn more about our cell culture proteins and technical services.
Email: product@eastmab.com
Phone: +86-400-998-0106