Recombinant Human Fibronectin: A Critical ECM Protein for Advanced Cell Culture and Regenerative Medicine
In modern cell culture and regenerative medicine, consistency is everything. As research moves toward serum-free systems, scalable stem cell manufacturing, and clinically relevant culture conditions, recombinant human fibronectin has become an increasingly important extracellular matrix (ECM) component for supporting reliable cellular behavior.
Unlike plasma-derived fibronectin, recombinant human fibronectin offers defined composition, superior batch-to-batch consistency, and scalable manufacturing under controlled conditions. These advantages make it an essential tool for researchers and biomanufacturers working in stem cell culture, tissue engineering, organoid development, and cell therapy applications.
What Is Recombinant Human Fibronectin?
Fibronectin is a high-molecular-weight glycoprotein naturally found in the extracellular matrix and blood plasma. It plays a central role in regulating:
Cell adhesion
Cell migration
Cell proliferation
Cell differentiation
Tissue repair and organization
Through interactions with integrins and other ECM molecules, fibronectin helps cells attach to surfaces and respond to their surrounding microenvironment.
Recombinant human fibronectin is a bioengineered form of this protein produced using controlled recombinant expression systems. Compared with plasma-derived sources, recombinant production enables:
Higher purity
Reduced contamination risks
Animal-origin free manufacturing options
Improved reproducibility
Scalable industrial production
These features are increasingly important in advanced research and translational applications where experimental consistency directly impacts data quality and process reliability.
Biological Functions of Recombinant Human Fibronectin
The multifunctional structure of fibronectin contains several binding domains that interact with:
Integrins
Collagen
Fibrin
Heparin
Growth factors
These interactions regulate critical cellular signaling pathways involved in tissue development, wound healing, and extracellular matrix remodeling.
In cell culture systems, recombinant human fibronectin creates a biologically active surface that supports robust cell attachment and spreading. Cells cultured on fibronectin-coated surfaces often demonstrate improved morphology, enhanced proliferation, and more stable phenotypes under defined culture conditions.
Applications of Recombinant Human Fibronectin
Stem Cell Culture
Recombinant human fibronectin is widely used in stem cell research and manufacturing, particularly for:
MSC culture
iPSC expansion
ESC maintenance
Primary cell culture
As serum-free and xeno-free systems become standard in regenerative medicine workflows, fibronectin provides a defined ECM substrate that supports stable cell growth while minimizing variability introduced by undefined animal-derived components.
In mesenchymal stem cell (MSC) culture, fibronectin promotes strong adhesion and supports sustained proliferation across multiple passages.
Serum-Free and Defined Cell Culture Systems
Researchers increasingly rely on recombinant human fibronectin in serum-free culture systems where cell attachment becomes more challenging without serum-derived adhesion molecules.
Fibronectin-coated surfaces help maintain:
Cell viability
Uniform morphology
Consistent proliferation
Improved experimental reproducibility
These advantages are particularly important in bioprocess development, high-throughput screening, and GMP-oriented manufacturing workflows.
Tissue Engineering and Regenerative Medicine
In tissue engineering applications, recombinant human fibronectin is commonly used to functionalize biomaterials and scaffold surfaces.
Fibronectin enhances:
Cell-scaffold integration
Cellular infiltration
Tissue organization
Biomaterial biocompatibility
By mimicking aspects of the native extracellular matrix, fibronectin helps create microenvironments that support tissue formation and regenerative processes.
Organoid and 3D Culture Applications
As organoid technologies continue advancing, ECM proteins such as recombinant human fibronectin are becoming increasingly important for establishing physiologically relevant 3D culture systems.
Fibronectin can support:
Organoid attachment
Structural organization
Cell-ECM signaling
Long-term culture stability
Combined with growth factors such as EGF, Noggin, Wnt3a, and R-spondin 1, recombinant fibronectin contributes to more defined and reproducible organoid culture platforms.
Why Quality Matters
The quality of recombinant human fibronectin directly affects experimental outcomes.
Impurities, endotoxins, or inconsistent protein activity can alter:
Cell attachment
Cell signaling
Differentiation efficiency
Morphology
Long-term culture stability
High-quality recombinant proteins reduce these risks and improve reproducibility across experiments and manufacturing batches.
At EastMabBio, recombinant human fibronectin is manufactured under rigorous quality control standards to support advanced research and translational applications.
Key features include:
High purity
Low endotoxin
High bioactivity
Batch-to-batch consistency
Scalable production capacity
Bioactivity Validation of EastMabBio Fibronectin (Y13142H)
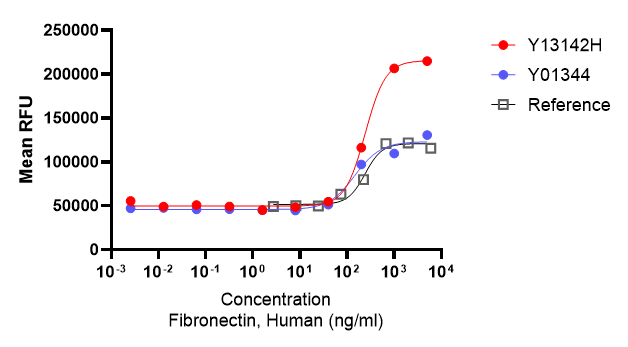
The activity of EastMabBio Fibronectin (Y13142H) is determined by its ability to support MSC adhesion when immobilized, withan ED50≤ 0.3 μg/mL.
This high bioactivity enables efficient cell attachment and supports robust MSC expansion under defined culture conditions.
Recombinant Human Fibronectin for the Future of Cell Manufacturing
As regenerative medicine, cell therapy, and organoid technologies continue evolving, demand for defined extracellular matrix proteins will continue to grow.
Recombinant human fibronectin provides:
Reliable cell adhesion
Defined culture conditions
Improved reproducibility
Scalable manufacturing compatibility
Support for serum-free culture
For researchers and biomanufacturers seeking more consistent and translationally relevant cell culture systems, recombinant human fibronectin has become an essential component of next-generation cell manufacturing platforms.