The CGT Era: When Cells and Genes Become "Living Drugs"
With the continuous advancement of life sciences and biotechnological engineering, the traditional treatment system centered on small molecule drugs and antibodies is being supplemented and reshaped by a new generation of therapies. Among them, cell and gene therapy (CGT), represented by immunotherapy, stem cell therapy, and gene therapy, has achieved a leap from "symptom relief" to "mechanism intervention" and even "disease cure" by directly regulating cell functions or modifying genetic information, and is gradually becoming an important development direction of modern medicine.
Immunocyte therapy: Precise regulation of the immune system
Immunotherapy involves the in vitro modification and expansion of immune cells, which are then reinfused into the body to enhance the recognition and elimination of diseased cells. Among them, CAR-T and TCR-T are the two most mature technologies in application.
CAR-T: It does not rely on MHC to recognize tumor antigens and has shown remarkable efficacy in hematological malignancies such as B-ALL and DLBCL.
TCR-T: By enhancing T cells' recognition of tumor-associated antigens, it holds greater potential in solid tumors.
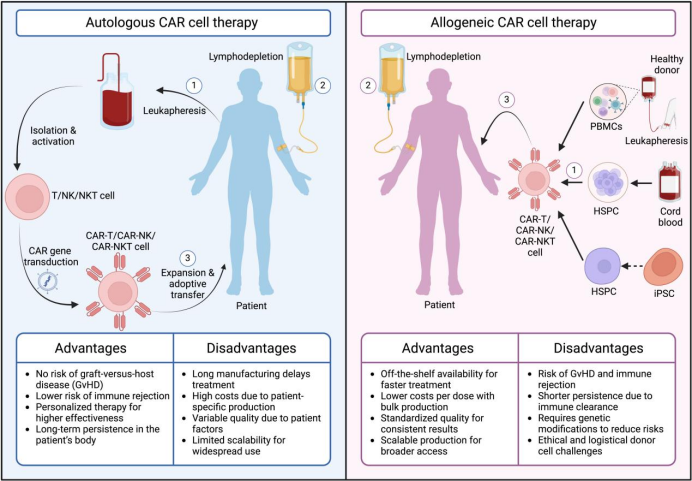
Comparison of Autologous and Allogeneic CAR-T Therapies (Image source: Med)
Among them, cytokines play a key regulatory role in the expansion and functional maintenance of immune cells:
IL-2: Promotes rapid proliferation of T cells, but high doses may accelerate differentiation.
IL-7/IL-15: Maintain the phenotype of memory T cells and enhance persistence
IL-21: Regulating differentiation and enhancing anti-tumor activity
Optimizing the combination and dosage of cytokines is a core link in determining the quality of cell products and clinical efficacy.
Stem cell therapy: Driving regenerative medicine and tissue repair
Stem cells possess the potential for self-renewal and multi-directional differentiation. Mesenchymal stem cells (MSCs) and induced pluripotent stem cells (iPSCs) are the two most actively studied and transformed types.
MSCs: With a wide range of sources (such as bone marrow, umbilical cord, etc.), they possess the ability of immune regulation and tissue repair.
iPSC: Acquiring pluripotency through somatic cell reprogramming, supporting personalized treatment and disease model construction.
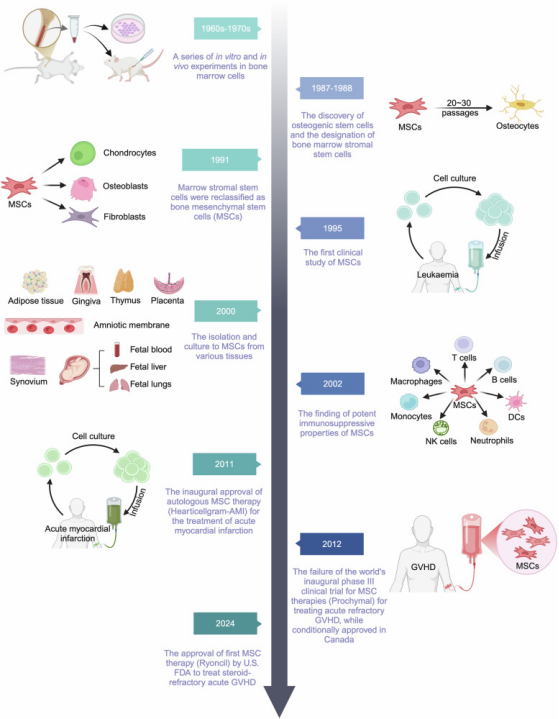
Research history and milestones of MSCs and related treatments (Image source: Signal Transduct Target Ther)
The main challenges: pluripotency maintenance, spontaneous differentiation control, genetic stability, batch consistency and large-scale amplification.
Key factor support system:
Growth factors (such as bFGF, EGF, TGF-β, etc.) regulate proliferation and differentiation.
Extracellular matrix proteins (such as Laminin, Fibronectin, Vitronectin, etc.) provide adhesion and signaling support.
Transferrin, albumin and other substances maintain the stability of the microenvironment.
With the promotion of serum-free and animal-component-free systems, recombinant proteins have become the foundation for the standardization of stem cell culture and its clinical translation.
Gene Therapy: From Repairing Genetic Defects to Precision Medicine
Gene therapy corrects genetic abnormalities at the source by introducing, replacing or editing genes. The development of viral vectors (such as AAV and lentivirus) and gene editing tools (such as CRISPR/Cas) has driven breakthroughs in the treatment of genetic diseases and tumors.
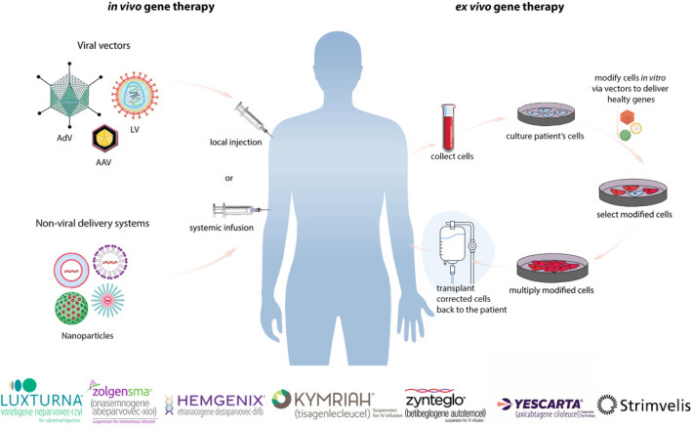
Differences between in vivo and in vitro gene therapy (Image source: J Cell Mol Med)
Core elements:
An efficient and safe delivery system
Precise gene editing capability
Controllable expression regulation mechanism
Gene therapy is often integrated with cell therapy (for instance, CAR-T is essentially an immune cell modified by gene), further expanding the application scope of CGT. Currently, challenges such as off-target effects, immunogenicity and long-term safety still need to be addressed.
Industrialization trend: Challenges of standardization and scale expansion
As CGT products enter clinical trials and the market, the industry is moving from technology validation to industrialization. The overall requirements for upstream raw materials are becoming increasingly strict:
High purity and low endotoxin content ensure cell function and safety.
Stable batch consistency to ensure product reproducibility
Clear bioactivity verification system to guarantee reliable protein function
GMP-compliant production and quality system
Improve regulatory support documents (such as DMF) to enhance the efficiency of application submission.
The development of automation and large-scale amplification technologies also places higher demands on the stable supply of raw materials and their scalability.
EastMabBio: GMP-grade Recombinant Proteins Supporting the Industrialization of CGT
EastMabBiooffers high-quality GMP-grade recombinant proteins and extracellular matrix products, comprehensively supporting the application and implementation of CGT from research and development to clinical use.
Immunocyte therapy
IL-2, IL-7, IL-15, and IL-21 —— Support the expansion and functional regulation of NK/T cells
Stem Cells and Regenerative Medicine
bFGF, EGF, IGF-1, TGF-β, Activin A, Transferrin, etc. —— Support stem cell expansion and fate regulation
Organoids and Tissue Engineering
Noggin, R-spondin 1, and Wnt-related factors - Building a stable organoid culture system
Extracellular matrix proteins
Laminin 511/521, Fibronectin, Vitronectin - Enhance cell adhesion, growth and functional performance
References
1. Li YR, Zhu Y, Fang Y, Lyu Z, Yang L. Emerging trends in clinical allogeneic CAR cell therapy. Med. 2025; 6(8):100677.
2. Han X, Liao R, Li X, et al. Mesenchymal stem cells in treating human diseases: molecular mechanisms and clinical studies.Signal Transduct Target Ther. 2025; 10(1):262.
3. Cetin B, Erendor F, Eksi YE, Sanlioglu AD, Sanlioglu S. Gene and cell therapy of human genetic diseases: Recent advances and future directions.J Cell Mol Med. 2024; 28(17):e70056.
News