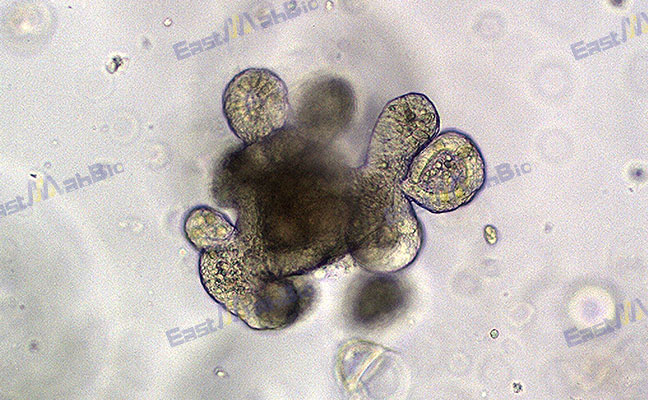
Gastric Organoids: Advancing Stomach Disease Research
Our work in biotechnology keeps showing how far advanced biological models can take us. Gastric organoids, miniature 3D tissue structures grown in vitro, have become a clear step forward. They closely mirror the stomach’s cellular architecture and physiological functions, letting us study gastric diseases and develop new therapies with a level of precision that traditional approaches rarely offer.
Understanding Gastric Organoids
Gastric organoids are self-organizing, three-dimensional cellular structures derived from stem cells. They recapitulate the cellular diversity and architectural complexity of the stomach. Organoid technology has become a powerful tool for disease modeling and drug screening.
What are Gastric Organoids
Gastric organoids are miniature stomach-like structures grown in a laboratory. They preserve key features of the native gastric epithelium, including multiple cell types and glandular organization. By offering models that are more physiologically relevant than traditional 2D cell cultures, organoid technology has reshaped in vitro research.
How Gastric Organoids are Developed
Gastric organoids are developed from specific stem cell sources.
1. We obtain stem cells from either induced pluripotent stem cells (iPSCs) or adult gastric tissue.
2. These cells are embedded within an extracellular matrix, typically Matrigel, which provides structural support.
3. Specific growth factors, including EGF (Cat#Y00801),Wnt3a (Cat#Y04603), Noggin (Cat#Y03301), R-spondin 1 (Cat#Y03501),
and FGF-10(Cat#Y01101), are added to the culture medium.
4. These factors guide the differentiation and self-organization of the stem cells into organoid structures.
5. The resulting organoids mature over several weeks, developing complex glandular structures.
Applications of Gastric Organoids in Research and Medicine
Gastric organoids serve as invaluable tools across various biomedical applications. They offer a robust platform for studying disease mechanisms and evaluating therapeutic interventions.
Modeling Gastric Diseases
Gastric organoids are extensively used to model a range of stomach diseases. We utilize them to investigate gastric cancer, observing tumor initiation and progression. They also support studies on Helicobacter pyloriinfection, clarifying host-pathogen interactions. Organoids additionally provide models for inflammatory bowel diseases, revealing insights into chronic inflammation. These models hold clear advantages over traditional animal models and 2D cell lines because of their physiological relevance.
Drug Discovery and Screening
High-throughput screening platforms integrate gastric organoids for efficient drug discovery. We employ these systems to identify novel therapeutic targets. Organoids allow us to test drug efficacy and assess potential toxicity in a more human-relevant context. This accelerates the development of new pharmaceutical agents.
Personalized Medicine Approaches
Patient-derived gastric organoids (PDOs) are central to precision medicine. We generate these organoids from individual patient biopsies. This enables us to predict specific patient responses to various therapies. Treatment strategies can then be tailored for optimal outcomes, strengthening personalized care.
Advantages and Challenges of Gastric Organoid Technology
Feature | Gastric Organoids | 2D Cell Cultures | Animal Models |
Physiological Relevance | High (3D structure, multiple cell types) | Low (monolayer, limited complexity) | High (whole organism context) |
Disease Modeling | Excellent (human-specific responses) | Limited (simplified context) | Good (complex interactions, but species-specific) |
Drug Screening | High-throughput, human-relevant | High-throughput, but less relevant | Low-throughput, ethical concerns |
Ethical Concerns | Low | Low | High |
Cost & Time | Moderate | Low | High |
Gastric organoid technology offers numerous benefits but also faces several limitations. Addressing these challenges will further expand their utility in research and clinical settings.
Benefits of Using Gastric Organoids
Gastric organoids provide significant advantages over conventional models. They show strong physiological relevance, accurately recapitulating tissue architecture and function. Ethical concerns are reduced compared to animal models, aligning with modern research principles. Organoids also scale well, fitting both academic research and industrial applications.
Current Limitations and Future Directions
Despite their advantages, gastric organoids face limitations. Achieving full vascularization and innervation remains challenging and is important for long-term maturation. Standardization of culture protocols is still in progress to improve reproducibility across laboratories. Emerging technologies, including microfluidics and advanced biomaterials, are being developed to address these limitations.
The Future of Gastric Organoids in Biomedical Research
The integration of gastric organoids with advanced ‘omics’ technologies will deepen our understanding of disease. We anticipate a growing role in regenerative medicine and tissue engineering, with the potential to enable new therapeutic interventions. High-quality recombinant protein raw materials are indispensable for advancing this work. They support consistent, robust organoid growth and help drive future discoveries.
Partner with EastMabBio for Your Research Needs
Explore EastMabBio’s high-quality recombinant protein raw materials, indispensable for advanced gastric organoid research and development. Contact us today to learn how our products can support your scientific endeavors.
Email: product@eastmab.com
Phone: +86-400-998-0106